Bioactive Lipid Mediators Core
Overview
The Bioactive Lipid Mediators Core (BLMC) is designed to support the ongoing work of scientists aiming to understand the systemic and neurophysiological outcomes of chronic drug use on lipid signaling (see C3A Mission statement). The focus on lipid biomarkers targets an emerging field that can add a novel framework for understanding how lipid signaling in a wide range of tissues (e.g., plasma, CNS, liver, breast milk) is related to homeostatic dysregulation often associated with the development and maintenance of substance use disorders.
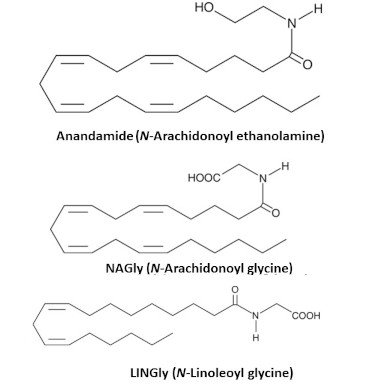
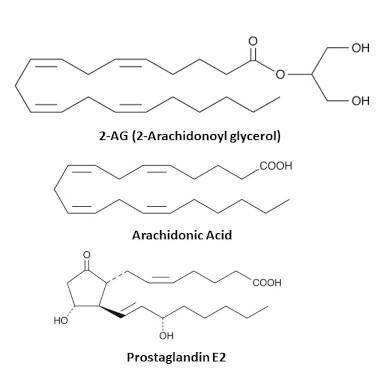
The Bradshaw lab has developed lipid extraction and analytical techniques aimed specifically at small molecule lipid like the endocannabinoids, Anandamide and 2-arachidonoyl glycerol, and their congeners, lipo amino acids, prostaglandins, leukotrienes, and resolvins among others. These classes of lipids can change in response to exposure to drugs such as cannabinoids and opioids and understanding their regulation has the potential to provide unique insights into how both acute and chronic drug use drives changes in systemic and CNS physiology. These insights would then help guide the discovery of novel therapies to treat addiction disorders.
Guidelines for BLMC Applications
Purpose
To determine general scientific scope match to the C3A mission and feasibility of study design for lipidomics analysis that will provide the best chance optimization for informative data. This is not an application that determines overall “scientific merit” and “innovation”. The focus here is on programmatic fit and study feasibility. BLMC analyses can complement ongoing, funded research; however, it is also understood that some projects are aimed at generating pilot data for future proposals.
Key Components
- Study relationship to the C3A mission (see above).
- General study design including subject type, number, genetic sex (if applicable), and tissue type(s) for analysis.
General Study Design Requirements
- All studies must have a baseline/control group that is an age, sex, and tissue type match with study target. (e.g. vehicle injection, wild-type control).
- For animal studies, typical Power analysis for most tissue types is a minimum number (n) of 6 and a maximum n of 15. All animal subject group n sizes must fall within this range and a brief rationale for the n must be provided (e.g. larger n for behavioral studies, smaller n for drug injection studies).
- For Human studies, every effort must be made to have equivalent control groups; however, these may have a wider range of conditions and variables than animal studies (e.g. only drug A and not drugs A and B, drug A with and without CBT, BMI ranges). Given that the tissue type for human subjects is largely limited to plasma and diet and time of day have effects on lipid content of plasma, an initial Power analysis of subject groups may be required prior to a larger-scale study acceptance. Indicate the rationale for the suggested group n in the application.
Acknowledgements
Disclaimer
Research and content found in this publication is supported by the National Institute On Drug Abuse of the National Institutes of Health under Award Number P30DA056410. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.

